Gene
KWMTBOMO00487 Validated by peptides from experiments
Pre Gene Modal
BGIBMGA012304
Annotation
1-Cys_peroxiredoxin_[Bombyx_mori]
Full name
Peroxiredoxin-6
Alternative Name
1-Cys peroxiredoxin
Acidic calcium-independent phospholipase A2
Antioxidant protein 2
Ciliary body glutathione peroxidase
Non-selenium glutathione peroxidase
PHGPx
Acidic calcium-independent phospholipase A2
Antioxidant protein 2
Ciliary body glutathione peroxidase
Non-selenium glutathione peroxidase
PHGPx
Location in the cell
Cytoplasmic Reliability : 2.106
Sequence
CDS
ATGCTTTTACTTGGAAAAACTTTTCCAGACTTCTCTGCCAACACAACAGAAGGCGAAATAAATTTTCACGAGTGGCTAGGAGATAAGTGGGGTATACTGTTCTCTCACCCATCAGACTTCACCCCGGTCTGCACAACAGAATTGGCTCGGGTGCTCGTCCTTCTTCCAGAGTTTGTGAAGCGCAACACAAAAGTCATTGGCCTGTCCTGTGACAGTGTATCCTCTCACTTGGAATGGTGCAAGGACATTAAGTCTTTTGCTGGTTGCAATGAAGACGAACCATTTCCTTATCCGATAATTGAAGACGAGAAGAGAGAGTTGGCCAACAAGCTCGGCATGATTGACAATGACGAATTGGATCACAAAGGAATGCCGCTGACAGCCCGTGCAGTCTTCATTGTCGATCCGAATAAGAAGTTTAGACTGTCAATATTGTATCCGGCTACGACTGGACGTAATTTTGATGAGATTCTGCGCATACTGGACTCTCTCCAGTTGACTGATAAGGCTAAAGTGGCAACGCCGGTAGATTGGAAGGCGGGTGACGACTGCATGGTGTTGCCAACCGTGCCGGAGGACCAGATCAAAACGTGTTTCCCACAGGGTGTCAACGTTGTTCCGCTTCCGTCCGGCAAGAATTATCTGAGGAAGACTGCTTGCCCTAAAATTTGA
Protein
MLLLGKTFPDFSANTTEGEINFHEWLGDKWGILFSHPSDFTPVCTTELARVLVLLPEFVKRNTKVIGLSCDSVSSHLEWCKDIKSFAGCNEDEPFPYPIIEDEKRELANKLGMIDNDELDHKGMPLTARAVFIVDPNKKFRLSILYPATTGRNFDEILRILDSLQLTDKAKVATPVDWKAGDDCMVLPTVPEDQIKTCFPQGVNVVPLPSGKNYLRKTACPKI
Summary
Description
Thiol-specific peroxidase that catalyzes the reduction of hydrogen peroxide and organic hydroperoxides to water and alcohols, respectively. Can reduce H(2)O(2) and short chain organic, fatty acid, and phospholipid hydroperoxides. Also has phospholipase activity, and can therefore either reduce the oxidized sn-2 fatty acyl grup of phospholipids (peroxidase activity) or hydrolyze the sn-2 ester bond of phospholipids (phospholipase activity). These activities are dependent on binding to phospholipids at acidic pH and to oxidized phospholipds at cytosolic pH. Plays a role in cell protection against oxidative stress by detoxifying peroxides and in phospholipid homeostasis.
Catalytic Activity
[protein]-dithiol + a hydroperoxide = [protein]-disulfide + an alcohol + H2O
a 1,2-diacyl-sn-glycero-3-phosphocholine + H2O = a 1-acyl-sn-glycero-3-phosphocholine + a fatty acid + H(+)
a 1,2-diacyl-sn-glycero-3-phosphocholine + H2O = a 1-acyl-sn-glycero-3-phosphocholine + a fatty acid + H(+)
Biophysicochemical Properties
25 uM for H(2)O(2)
180 uM for H(2)O(2)
22 uM for tert-butyl hydroperoxide
142 uM for tert-butyl hydroperoxide
170 uM for cumene hydroperoxide
120 uM for cumene hydroperoxide
12 uM for triphenylcarbinyl hydroperoxide
34 uM for linoleic hydroperoxide
141 uM for linolenoyl hydroperoxide
135 uM for arachidonoyl hydroperoxide
120 uM for PLCP hydroperoxide
129 uM for PACP hydroperoxide
22 uM for 5-phenyl-3-pentenyl hydroperoxide
350 uM for dipalmitoyl phosphatidylcholine (at pH 4)
180 uM for H(2)O(2)
22 uM for tert-butyl hydroperoxide
142 uM for tert-butyl hydroperoxide
170 uM for cumene hydroperoxide
120 uM for cumene hydroperoxide
12 uM for triphenylcarbinyl hydroperoxide
34 uM for linoleic hydroperoxide
141 uM for linolenoyl hydroperoxide
135 uM for arachidonoyl hydroperoxide
120 uM for PLCP hydroperoxide
129 uM for PACP hydroperoxide
22 uM for 5-phenyl-3-pentenyl hydroperoxide
350 uM for dipalmitoyl phosphatidylcholine (at pH 4)
Subunit
Homodimer (By similarity). Interacts with GSTP1; mediates PRDX6 glutathionylation and regeneration (PubMed:15004285). Interacts with APEX1. Interacts with STH. May interact with FAM168B (By similarity). May interact with HTR2A (By similarity).
Homodimer (By similarity). Interacts with GSTP1; mediates PRDX6 glutathionylation and regeneration (By similarity).
Homodimer (By similarity). Interacts with GSTP1; mediates PRDX6 glutathionylation and regeneration (By similarity).
Miscellaneous
The active site is a conserved redox-active cysteine residue, the peroxidatic cysteine (C(P)), which makes the nucleophilic attack on the peroxide substrate. The peroxide oxidizes the C(P)-SH to cysteine sulfenic acid (C(P)-SOH), which then reacts with another cysteine residue, the resolving cysteine (C(R)), to form a disulfide bridge. The disulfide is subsequently reduced by an appropriate electron donor to complete the catalytic cycle. In this 1-Cys peroxiredoxin, no C(R) is present and C(P) instead forms a disulfide with a cysteine from another protein or with a small thiol molecule. C(P) is reactivated by glutathionylation mediated by glutathione S-transferase Pi, followed by spontaneous reduction of the enzyme with glutathione.
Similarity
Belongs to the peroxiredoxin family. Prx6 subfamily.
Keywords
Acetylation
Antioxidant
Complete proteome
Cytoplasm
Direct protein sequencing
Hydrolase
Lipid degradation
Lipid metabolism
Lysosome
Multifunctional enzyme
Oxidoreductase
Peroxidase
Phosphoprotein
Redox-active center
Reference proteome
Feature
chain Peroxiredoxin-6
Uniprot
A8D0B7
Q2F6C8
A0A212EQF8
S4NJ02
A0A2H1WM86
A0A2A4J269
+ More
A0A194QZI0 A0A2S0RQR9 T1TCE9 U5ET43 A0A182Y299 A0A1Y1N188 A0A1L8E456 A0A182L3E2 D6WJ53 A0A182WWP6 A0A182M6M9 Q7QIQ4 A0A182I6Y2 A0A182PF44 A0A182V5P2 A0A182TU07 A0A182QC30 A0A1B6FZH1 A0A2M4BYL2 A0A0K8TS29 A0A2M4BYQ0 A0A182R343 A0A182JXG7 A0A182FDP3 W8CC17 A0A1J1J5H9 A0A182WL43 B5G0S1 A0A218V5B1 V5GAQ6 T1DJM5 A0A084W9Y0 A0A1B6BZ20 A0A1B6KS88 A0A0C9R5D5 W5JFL0 A0A182NJL3 U3JMG6 A0A2M4AX40 A0A336LYC4 A0A336M4R2 B5G0S3 A0A3M0K533 A0A2M4AX00 A0A0K8US52 A0A026X1B2 A0A034VZ73 A0A0A1WIE6 A0A2L2Y558 A0A182IXP6 W8Q918 A0A182YTR2 F6RBB7 E2C0C6 A0A088AKM8 N6U1R2 J3JW46 A0A2H4GVH0 A0A2Y9F3L9 E0VEQ3 A0A2I0MC11 K4FY71 H9CTT2 A0A2I7NB29 A0A2U3WK92 A0A1B6HC28 W5PQA8 A0A226MRT1 A0A226PVT3 A0A341ADK1 A0A2Y9MUT0 L8IXN6 M4T8F1 A0A212CR39 O77834 A0A2U3XU27 K4G5J3 A0A340WI73 A0A383YRH6 A0A2Y9GGK9 F1NBV0 A0A2P4TBV4 F1PC59 A0A158NH13 K4G7M7 A0A2I2UL25 A0A195B6G1 M3XZW0 A0A2Y9JSS2 A0A1V4JYA8 Q5ZJF4 A0A369S9G8 B3RP43 A0A1S3F3K8
A0A194QZI0 A0A2S0RQR9 T1TCE9 U5ET43 A0A182Y299 A0A1Y1N188 A0A1L8E456 A0A182L3E2 D6WJ53 A0A182WWP6 A0A182M6M9 Q7QIQ4 A0A182I6Y2 A0A182PF44 A0A182V5P2 A0A182TU07 A0A182QC30 A0A1B6FZH1 A0A2M4BYL2 A0A0K8TS29 A0A2M4BYQ0 A0A182R343 A0A182JXG7 A0A182FDP3 W8CC17 A0A1J1J5H9 A0A182WL43 B5G0S1 A0A218V5B1 V5GAQ6 T1DJM5 A0A084W9Y0 A0A1B6BZ20 A0A1B6KS88 A0A0C9R5D5 W5JFL0 A0A182NJL3 U3JMG6 A0A2M4AX40 A0A336LYC4 A0A336M4R2 B5G0S3 A0A3M0K533 A0A2M4AX00 A0A0K8US52 A0A026X1B2 A0A034VZ73 A0A0A1WIE6 A0A2L2Y558 A0A182IXP6 W8Q918 A0A182YTR2 F6RBB7 E2C0C6 A0A088AKM8 N6U1R2 J3JW46 A0A2H4GVH0 A0A2Y9F3L9 E0VEQ3 A0A2I0MC11 K4FY71 H9CTT2 A0A2I7NB29 A0A2U3WK92 A0A1B6HC28 W5PQA8 A0A226MRT1 A0A226PVT3 A0A341ADK1 A0A2Y9MUT0 L8IXN6 M4T8F1 A0A212CR39 O77834 A0A2U3XU27 K4G5J3 A0A340WI73 A0A383YRH6 A0A2Y9GGK9 F1NBV0 A0A2P4TBV4 F1PC59 A0A158NH13 K4G7M7 A0A2I2UL25 A0A195B6G1 M3XZW0 A0A2Y9JSS2 A0A1V4JYA8 Q5ZJF4 A0A369S9G8 B3RP43 A0A1S3F3K8
EC Number
1.11.1.15
Pubmed
17933572
19121390
22118469
23622113
26354079
25244985
+ More
28004739 20966253 18362917 19820115 12364791 14747013 17210077 26369729 24495485 17018643 24438588 20920257 23761445 24508170 30249741 25348373 25830018 26561354 24747012 24389255 15562597 18464734 20798317 23537049 22516182 20566863 23371554 23056606 29326594 20809919 24621616 22751099 25712755 9748299 10409692 16305752 2373154 9787801 15004285 15592404 16341006 21347285 17975172 15642098 30042472 18719581
28004739 20966253 18362917 19820115 12364791 14747013 17210077 26369729 24495485 17018643 24438588 20920257 23761445 24508170 30249741 25348373 25830018 26561354 24747012 24389255 15562597 18464734 20798317 23537049 22516182 20566863 23371554 23056606 29326594 20809919 24621616 22751099 25712755 9748299 10409692 16305752 2373154 9787801 15004285 15592404 16341006 21347285 17975172 15642098 30042472 18719581
EMBL
BABH01016932
BABH01016933
BABH01016934
BABH01016935
EU128675
ABV32570.1
+ More
DQ311144 ABD36089.1 AGBW02013269 OWR43730.1 GAIX01013874 JAA78686.1 ODYU01009260 SOQ53554.1 NWSH01003565 PCG66101.1 KQ461108 KPJ09001.1 MF979110 AWA45973.1 KC996761 AGT62471.1 GANO01002097 JAB57774.1 GEZM01015464 GEZM01015463 JAV91629.1 GFDF01000554 JAV13530.1 KQ971342 EFA04630.1 AXCM01003872 AAAB01008807 EAA04146.2 APCN01003711 AXCN02000440 GECZ01014184 JAS55585.1 GGFJ01008999 MBW58140.1 GDAI01000446 JAI17157.1 GGFJ01009001 MBW58142.1 GAMC01004291 JAC02265.1 CVRI01000070 CRL07210.1 DQ215156 DQ215157 DQ215159 EF191678 EF191685 ACH44882.1 MUZQ01000049 OWK60910.1 GALX01001261 JAB67205.1 GAMD01001371 JAB00220.1 ATLV01021970 KE525326 KFB47024.1 GEDC01031019 JAS06279.1 GEBQ01025664 JAT14313.1 GBYB01011519 JAG81286.1 ADMH02001540 ETN62128.1 AGTO01020768 GGFK01012045 MBW45366.1 UFQT01000285 SSX22800.1 UFQT01000393 SSX23923.1 DQ215158 ACH44884.1 QRBI01000117 RMC08302.1 GGFK01012002 MBW45323.1 GDHF01022792 GDHF01006648 JAI29522.1 JAI45666.1 KK107063 QOIP01000005 EZA61154.1 RLU22577.1 GAKP01010341 JAC48611.1 GBXI01015635 GBXI01012416 JAC98656.1 JAD01876.1 IAAA01001924 LAA03316.1 KF745893 KF745894 KZ288285 AHL44194.1 AIR96016.1 PBC29460.1 GL451770 EFN78585.1 APGK01045856 KB741039 KB632188 ENN74546.1 ERL89793.1 BT127464 AEE62426.1 KY204028 AQW41375.1 DS235093 EEB11859.1 AKCR02000021 PKK27212.1 JX052789 AFK11017.1 JQ364950 AFD04441.1 MF467270 AUR53651.1 GECU01035475 GECU01034886 JAS72231.1 JAS72820.1 AMGL01022133 MCFN01000500 OXB57991.1 AWGT02000005 OXB84226.1 JH880647 ELR59937.1 KC342239 AGH70289.1 MKHE01000014 OWK08382.1 AF080228 AF090194 AJ243848 BT020967 BC102172 JX208657 AFM86971.1 AADN05000934 PPHD01002801 POI33850.1 AAEX03005208 ADTU01015424 JX209226 AFM87540.1 AANG04002195 KQ976574 KYM80096.1 AEYP01054466 AEYP01054467 LSYS01005497 OPJ77131.1 AJ720480 NOWV01000038 RDD43480.1 DS985242 EDV27570.1
DQ311144 ABD36089.1 AGBW02013269 OWR43730.1 GAIX01013874 JAA78686.1 ODYU01009260 SOQ53554.1 NWSH01003565 PCG66101.1 KQ461108 KPJ09001.1 MF979110 AWA45973.1 KC996761 AGT62471.1 GANO01002097 JAB57774.1 GEZM01015464 GEZM01015463 JAV91629.1 GFDF01000554 JAV13530.1 KQ971342 EFA04630.1 AXCM01003872 AAAB01008807 EAA04146.2 APCN01003711 AXCN02000440 GECZ01014184 JAS55585.1 GGFJ01008999 MBW58140.1 GDAI01000446 JAI17157.1 GGFJ01009001 MBW58142.1 GAMC01004291 JAC02265.1 CVRI01000070 CRL07210.1 DQ215156 DQ215157 DQ215159 EF191678 EF191685 ACH44882.1 MUZQ01000049 OWK60910.1 GALX01001261 JAB67205.1 GAMD01001371 JAB00220.1 ATLV01021970 KE525326 KFB47024.1 GEDC01031019 JAS06279.1 GEBQ01025664 JAT14313.1 GBYB01011519 JAG81286.1 ADMH02001540 ETN62128.1 AGTO01020768 GGFK01012045 MBW45366.1 UFQT01000285 SSX22800.1 UFQT01000393 SSX23923.1 DQ215158 ACH44884.1 QRBI01000117 RMC08302.1 GGFK01012002 MBW45323.1 GDHF01022792 GDHF01006648 JAI29522.1 JAI45666.1 KK107063 QOIP01000005 EZA61154.1 RLU22577.1 GAKP01010341 JAC48611.1 GBXI01015635 GBXI01012416 JAC98656.1 JAD01876.1 IAAA01001924 LAA03316.1 KF745893 KF745894 KZ288285 AHL44194.1 AIR96016.1 PBC29460.1 GL451770 EFN78585.1 APGK01045856 KB741039 KB632188 ENN74546.1 ERL89793.1 BT127464 AEE62426.1 KY204028 AQW41375.1 DS235093 EEB11859.1 AKCR02000021 PKK27212.1 JX052789 AFK11017.1 JQ364950 AFD04441.1 MF467270 AUR53651.1 GECU01035475 GECU01034886 JAS72231.1 JAS72820.1 AMGL01022133 MCFN01000500 OXB57991.1 AWGT02000005 OXB84226.1 JH880647 ELR59937.1 KC342239 AGH70289.1 MKHE01000014 OWK08382.1 AF080228 AF090194 AJ243848 BT020967 BC102172 JX208657 AFM86971.1 AADN05000934 PPHD01002801 POI33850.1 AAEX03005208 ADTU01015424 JX209226 AFM87540.1 AANG04002195 KQ976574 KYM80096.1 AEYP01054466 AEYP01054467 LSYS01005497 OPJ77131.1 AJ720480 NOWV01000038 RDD43480.1 DS985242 EDV27570.1
Proteomes
UP000005204
UP000007151
UP000218220
UP000053240
UP000076408
UP000075882
+ More
UP000007266 UP000076407 UP000075883 UP000007062 UP000075840 UP000075885 UP000075903 UP000075902 UP000075886 UP000075900 UP000075881 UP000069272 UP000183832 UP000075920 UP000197619 UP000030765 UP000000673 UP000075884 UP000016665 UP000269221 UP000053097 UP000279307 UP000075880 UP000242457 UP000076420 UP000002279 UP000008237 UP000005203 UP000019118 UP000030742 UP000248484 UP000009046 UP000053872 UP000245340 UP000002356 UP000198323 UP000198419 UP000252040 UP000248483 UP000009136 UP000245341 UP000265300 UP000261681 UP000248481 UP000000539 UP000002254 UP000005205 UP000011712 UP000078540 UP000000715 UP000248482 UP000190648 UP000253843 UP000009022 UP000081671
UP000007266 UP000076407 UP000075883 UP000007062 UP000075840 UP000075885 UP000075903 UP000075902 UP000075886 UP000075900 UP000075881 UP000069272 UP000183832 UP000075920 UP000197619 UP000030765 UP000000673 UP000075884 UP000016665 UP000269221 UP000053097 UP000279307 UP000075880 UP000242457 UP000076420 UP000002279 UP000008237 UP000005203 UP000019118 UP000030742 UP000248484 UP000009046 UP000053872 UP000245340 UP000002356 UP000198323 UP000198419 UP000252040 UP000248483 UP000009136 UP000245341 UP000265300 UP000261681 UP000248481 UP000000539 UP000002254 UP000005205 UP000011712 UP000078540 UP000000715 UP000248482 UP000190648 UP000253843 UP000009022 UP000081671
Interpro
SUPFAM
SSF52833
SSF52833
ProteinModelPortal
A8D0B7
Q2F6C8
A0A212EQF8
S4NJ02
A0A2H1WM86
A0A2A4J269
+ More
A0A194QZI0 A0A2S0RQR9 T1TCE9 U5ET43 A0A182Y299 A0A1Y1N188 A0A1L8E456 A0A182L3E2 D6WJ53 A0A182WWP6 A0A182M6M9 Q7QIQ4 A0A182I6Y2 A0A182PF44 A0A182V5P2 A0A182TU07 A0A182QC30 A0A1B6FZH1 A0A2M4BYL2 A0A0K8TS29 A0A2M4BYQ0 A0A182R343 A0A182JXG7 A0A182FDP3 W8CC17 A0A1J1J5H9 A0A182WL43 B5G0S1 A0A218V5B1 V5GAQ6 T1DJM5 A0A084W9Y0 A0A1B6BZ20 A0A1B6KS88 A0A0C9R5D5 W5JFL0 A0A182NJL3 U3JMG6 A0A2M4AX40 A0A336LYC4 A0A336M4R2 B5G0S3 A0A3M0K533 A0A2M4AX00 A0A0K8US52 A0A026X1B2 A0A034VZ73 A0A0A1WIE6 A0A2L2Y558 A0A182IXP6 W8Q918 A0A182YTR2 F6RBB7 E2C0C6 A0A088AKM8 N6U1R2 J3JW46 A0A2H4GVH0 A0A2Y9F3L9 E0VEQ3 A0A2I0MC11 K4FY71 H9CTT2 A0A2I7NB29 A0A2U3WK92 A0A1B6HC28 W5PQA8 A0A226MRT1 A0A226PVT3 A0A341ADK1 A0A2Y9MUT0 L8IXN6 M4T8F1 A0A212CR39 O77834 A0A2U3XU27 K4G5J3 A0A340WI73 A0A383YRH6 A0A2Y9GGK9 F1NBV0 A0A2P4TBV4 F1PC59 A0A158NH13 K4G7M7 A0A2I2UL25 A0A195B6G1 M3XZW0 A0A2Y9JSS2 A0A1V4JYA8 Q5ZJF4 A0A369S9G8 B3RP43 A0A1S3F3K8
A0A194QZI0 A0A2S0RQR9 T1TCE9 U5ET43 A0A182Y299 A0A1Y1N188 A0A1L8E456 A0A182L3E2 D6WJ53 A0A182WWP6 A0A182M6M9 Q7QIQ4 A0A182I6Y2 A0A182PF44 A0A182V5P2 A0A182TU07 A0A182QC30 A0A1B6FZH1 A0A2M4BYL2 A0A0K8TS29 A0A2M4BYQ0 A0A182R343 A0A182JXG7 A0A182FDP3 W8CC17 A0A1J1J5H9 A0A182WL43 B5G0S1 A0A218V5B1 V5GAQ6 T1DJM5 A0A084W9Y0 A0A1B6BZ20 A0A1B6KS88 A0A0C9R5D5 W5JFL0 A0A182NJL3 U3JMG6 A0A2M4AX40 A0A336LYC4 A0A336M4R2 B5G0S3 A0A3M0K533 A0A2M4AX00 A0A0K8US52 A0A026X1B2 A0A034VZ73 A0A0A1WIE6 A0A2L2Y558 A0A182IXP6 W8Q918 A0A182YTR2 F6RBB7 E2C0C6 A0A088AKM8 N6U1R2 J3JW46 A0A2H4GVH0 A0A2Y9F3L9 E0VEQ3 A0A2I0MC11 K4FY71 H9CTT2 A0A2I7NB29 A0A2U3WK92 A0A1B6HC28 W5PQA8 A0A226MRT1 A0A226PVT3 A0A341ADK1 A0A2Y9MUT0 L8IXN6 M4T8F1 A0A212CR39 O77834 A0A2U3XU27 K4G5J3 A0A340WI73 A0A383YRH6 A0A2Y9GGK9 F1NBV0 A0A2P4TBV4 F1PC59 A0A158NH13 K4G7M7 A0A2I2UL25 A0A195B6G1 M3XZW0 A0A2Y9JSS2 A0A1V4JYA8 Q5ZJF4 A0A369S9G8 B3RP43 A0A1S3F3K8
PDB
1PRX
E-value=1.47921e-75,
Score=716
Ontologies
GO
GO:0005623
GO:0051920
GO:0045454
GO:0004601
GO:0034599
GO:0005829
GO:0042744
GO:0048471
GO:0042803
GO:0046475
GO:0006979
GO:0004602
GO:0048026
GO:0031625
GO:0047499
GO:0005634
GO:0005764
GO:0005737
GO:0004623
GO:0102567
GO:0000302
GO:0016042
GO:0102568
GO:0005615
GO:0016209
GO:0016491
GO:0030126
GO:0005488
GO:0003677
GO:0003707
GO:0007169
GO:0016021
GO:0055114
Topology
Subcellular location
Cytoplasm
Also found in lung secretory organelles (lamellar bodies). With evidence from 8 publications.
Lysosome Also found in lung secretory organelles (lamellar bodies). With evidence from 8 publications.
Lysosome Also found in lung secretory organelles (lamellar bodies). With evidence from 8 publications.
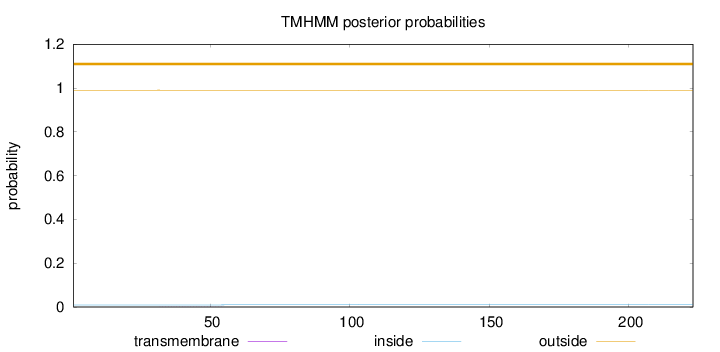
Length:
223
Number of predicted TMHs:
0
Exp number of AAs in TMHs:
0.00782
Exp number, first 60 AAs:
0.00782
Total prob of N-in:
0.00955
outside
1 - 223
Population Genetic Test Statistics
Pi
13.93521
Theta
16.214054
Tajima's D
-0.942041
CLR
0.304731
CSRT
0.145342732863357
Interpretation
Uncertain
